 Immuno-oncology agents, particularly checkpoint inhibitors, have moved to the forefront of cancer research and treatment in recent years. Indeed, the discovery that cancer could be treated by activating the immune system was honored with a Nobel Prize in 2018. But, as revolutionary as immuno-oncology drugs have been for the cancer field, many patients still do not respond well to these treatments as single agents. As a result, researchers have looked to a variety of combination approaches to boost the efficacy of these therapeutics.
Immuno-oncology agents, particularly checkpoint inhibitors, have moved to the forefront of cancer research and treatment in recent years. Indeed, the discovery that cancer could be treated by activating the immune system was honored with a Nobel Prize in 2018. But, as revolutionary as immuno-oncology drugs have been for the cancer field, many patients still do not respond well to these treatments as single agents. As a result, researchers have looked to a variety of combination approaches to boost the efficacy of these therapeutics.
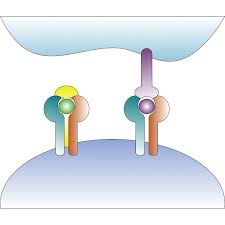
Many such efforts focus on using two or more agents together to treat cancer. Recently, however, increasing attention has been directed to a new generation of immuno-oncology agents — “fusion proteins” or bispecific antibodies that enable complex biological activities by simultaneously binding to two different antigens. Typically, these agents recognize a tumor antigen and a receptor on T cells, effectively bringing tumor and immune cells in close proximity. This offers a novel way to fight tumors that would otherwise escape immune regulation. While the concept of bispecific antibodies was invented more than 25 years ago, only in recent years have growing numbers of companies entered the space due to former difficulties engineering these molecules.
One of the current leaders in the field is Merck KGaA, who recently signed a $4.2 billion collaboration with GlaxoSmithKline focused on Merck’s bispecific, M7824. This molecule combines anti-PD-L1 activity with a TGF-β trap, and is designed to simultaneously block two immunosuppressive pathways to potentially restore and enhance anti-tumor responses. M7824 is currently in a mid-stage clinical trial versus Keytruda as a potential first-line treatment in PD-L1 advanced non-small cell lung cancer. It is also being explored in nine other solid tumor types including human HPV-associated cancers, biliary tract cancer, and gastric cancer — both on its own and in combination with other agents.
Clinical research has suggested that therapies based on immunostimulating cytokines, such as IL-15, could increase the efficacy of checkpoint inhibitors. Xencor has signed a partnership with Genentech focused on the development of XmAb24306, an engineered IL-15/IL-15Rα cytokine complex. Genentech is also developing its own bispecific agents, including mosunetuzumab, a molecule that targets both CD3 on T cells and CD20, a cell surface protein expressed by most malignant B cells, for enhanced B cell killing in non-Hodgkin’s lymphoma.
Amgen has also made bispecific antibodies a focus, and today they represent about 60% of the company’s oncology pipeline. One such agent, Blincyto (blinatumomab), was approved in 2014 to treat a rare type of acute lymphoblastic leukemia and is currently in clinical studies for other forms of leukemia. Amgen says its BiTE bi-specific antibody platform enables the company to quickly engineer bi-functional antibodies to a wide range of tumors, by taking the binding specificities against different surface antigens on malignant cells and linking them to the CD3-binding domain.