 Effective diabetes care is a complex, information-rich daily challenge. Besides lowering blood glucose levels, patients need to cope with the risk of hypoglycemia, manage the effects of their dietary choices and activity levels, and monitor the potential side-effects associated with their medications and possible related conditions, such as obesity and hypertension.
Effective diabetes care is a complex, information-rich daily challenge. Besides lowering blood glucose levels, patients need to cope with the risk of hypoglycemia, manage the effects of their dietary choices and activity levels, and monitor the potential side-effects associated with their medications and possible related conditions, such as obesity and hypertension.
Earlier this year, we wrote about the decision by the U.S. Food and Drug Administration to foster innovation in diabetes care through new rules and guidance on development pathways. This decision sought to lift barriers to the creation and commercialization of new devices and software capable of interacting and sharing data important to effective blood glucose monitoring and better overall diabetes care. In line with these policy changes, the agency approved Tandem Diabetes Care for the first truly interoperable insulin pump. The device was designed to securely and reliably communicate with any compatible external device to receive, execute and confirm drug delivery commands.
At that time, we asked whether other medical device developers would follow suit and ensure their diabetes management products helped create a more favorable ecosystem and a better experience for patients. The answer has been a resounding “Yes!” as several major collaborations between key players in the diabetes management field have emerged in recent weeks.
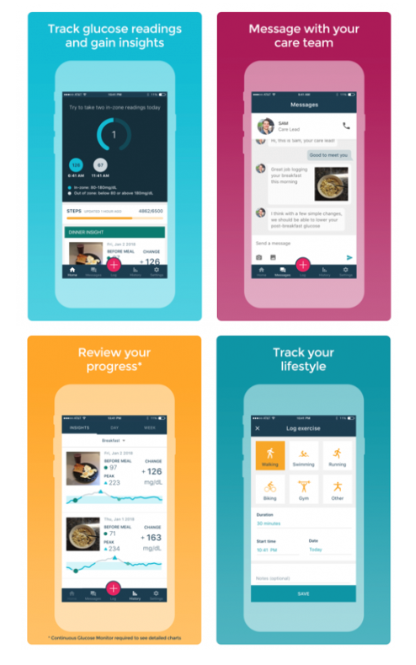
In September, Abbott and Sanofi announced a non-exclusive collaboration aimed at interconnecting the companies’ glucose monitoring and insulin delivery technologies. Their goal is to simplify patients’ experiences by facilitating data sharing and analytics via cloud-based solutions and smart-phone apps. The two companies are currently gathering user consent to digitally connect and share data between Abbott’s FreeStyle Libre wearable glucose sensor and Sanofi’s digital insulin pens.
Sanofi has been taking a leadership role in seeking to improve care for patients with diabetes through digital initiatives. The company previously launched a “virtual diabetes clinic” called Onduo in a joint venture with Verily. The Onduo digital platform connects monitoring /insulin delivery devices and software with personalized coaching and advice to offer a more patient-centric disease management experience. Sanofi, Verily, and Sensile Medical are also collaborating on the development of an internet-connected, all-in-one insulin patch and pump for patients with Type-2 diabetes — an important development, as most current insulin pump technology is specifically focused on Type 1 diabetes.
Similarly, Medtronic and Novo Nordisk announced a non-exclusive collaboration in September to integrate their diabetes-related offerings and share data between Medtronic’s continuous glucose monitoring devices and the smart insulin pens that Novo Nordisk plans to launch in 2020. Novo Nordisk has also signed data compatibility agreements with Abbott, Glooko and other companies.
Bayer also announced plans to work with start-up company OneDrop — and has invested in that company — to develop a Bluetooth-enabled blood glucose meter, test strips and mobile app that includes personalized coaching. OneDrop additionally communicates with Fitbit trackers and DexCom glucose monitoring systems, tying information from each source through a mobile platform that runs on iPhone and Android devices. Bayer is further planning to harness OneDrop’s platform across other therapeutic areas, initially looking at opportunities to improve patient care in oncology, heart disease and women’s health.
As these collaborations have so far been non-exclusive, will we continue to see diabetes players integrating their technology platforms with those of their competitors? Will other companies seeking to improve patient care through digital standardization and data compatibility seek to translate diabetes initiatives to other conditions requiring chronic disease management? Will further changes to FDA be required to address such developments? We’ll continue to watch this space.